Clean-in-place (CIP) procedures are a routine part of operating RO systems. Cleanings are typically initiated when normalized performance deviates beyond a defined threshold, such as a 10–15% decline in normalized permeate flow, an increase in differential pressure, or a change in salt passage. Once triggered, the process is straightforward: schedule the cleaning, apply the established protocol, and return the system to service.
But one critical question is often overlooked: how was that cleaning protocol chosen?
In many plants, CIP chemistry and sequencing are based on inherited procedures, past experience, or general manufacturer guidance. While these approaches may work at times, they often assume the type of foulant rather than confirm it. When the chemistry does not match the actual foulant, cleanings become less effective, intervals shorten, and membrane performance gradually declines.
Ineffective cleanings are costly. A train may be offline for a full day, resulting in lost production, chemical expense, and labor hours. At the same time, every CIP exposes membranes to extreme pH, chemical stress, and temperature. Although cleaning is necessary to prevent irreversible fouling, repeated exposure accelerates membrane aging. Extending membrane life is therefore not just about cleaning when needed; it is about cleaning correctly.
The goal is not to clean more often or use stronger chemicals. The goal is to minimize cleaning frequency by fully removing the foulant present. When deposits are only partially removed, recovery is incomplete and the next cleaning comes sooner than expected, even if pretreatment is operating properly. This happens because the same permeate flow must then be produced with less available membrane area. The resulting increase in flux across the remaining active surface forces more foulants toward the membrane surface, accelerating deposition and causing the system to foul again more quickly.
Selecting the right CIP protocol is not routine maintenance. It is a diagnostic process. It requires identifying what is actually on the membrane and validating that the chosen chemistry will remove it effectively. Only then can CIP move from reactive response to controlled, performance-driven operation.
Not All Fouling Responds the Same Way
There are two ways a foulant can be removed during a CIP: mechanically and chemically. Mechanical removal comes from flow. The higher the flow rate, the more shear force is applied across the membrane surface, which helps to scour and lift material off. Chemical removal happens when the cleaning solution dissolves or disperses the foulant so it can be flushed out of the system.
The problem is that we are limited mechanically. During a cleaning, we cannot exceed a certain differential pressure without risking membrane damage. In most systems, about 40–45 gpm per pressure vessel is the practical upper limit. That means we cannot rely on flow alone to remove deposits. For a cleaning to be effective, the chemistry must make the foulant soluble or dispersible under conditions the membrane can tolerate.
This is where a common misconception comes in. Fouling is often described as either “acid-soluble” or “caustic-soluble.” In reality, it is not that simple. Not every foulant responds strongly to pH, and even when it does, the pH required to dissolve it may be far outside the safe operating range for RO membranes. When cleaning chemistry is chosen based on assumptions, it can result in expensive cleanings that barely touch the foulant.
Take calcium carbonate as an example. It is strongly pH dependent. If antiscalant feed is interrupted and carbonate scale forms, a properly controlled low pH cleaning, typically around pH 2–3, can dissolve the scale and restore performance effectively.
Graphic 1: When acid reacts with calcium carbonate, the solid scale dissolves and releases carbon dioxide gas, which appears as bubbles. This is the same reaction that occurs during a properly controlled low-pH cleaning in an RO system.
Calcium sulfate, on the other hand, behaves very differently. It does not dissolve under normal acidic cleaning conditions, and it is only slightly more soluble at high pH. While it would dissolve in extremely strong caustic, those conditions would destroy an RO membrane. In practice, calcium sulfate often requires specialty chemistry designed to break apart the crystal structure so it can be flushed away.
Graphic 2: Calcium sulfate slurry formed after cleaning with a specialty chemical

Graphic 3: Due to severe scaling, flat-sheet testing was not possible. Feed spacers were used instead. Spacers were suspended in a beaker and agitated with a stir bar (1 minute, 3 times per day)
Silica is even more challenging. Once polymerized, silica forms a dense, glass-like layer. Although silica solubility increases at higher pH, the maximum pH that a membrane can safely tolerate is not high enough to significantly dissolve mature silica scale. High pH cleanings can also cause temporary membrane swelling, which may make permeability appear improved immediately after cleaning even if the silica was not removed. Once the membrane returns to normal, performance often declines again.

Graphic 4: Although permeability (specific flux) appeared to return to manufacturer specifications, the silica deposits remained. The apparent improvement was due to temporary membrane swelling, not true scale removal.
Effective silica removal requires chemistry that can break the structure itself, and if the scale is advanced, recovery may be limited. Fluoride-based cleaners are often used for this purpose because fluoride ions can react with the silica network and solubilize it. However, traditional fluoride sources such as hydrofluoric acid or ammonium bifluoride pose significant handling hazards, as they are highly corrosive and can cause severe chemical burns. For this reason, specialized formulated cleaners have been developed that incorporate stabilized fluoride chemistry designed to safely remove silica deposits while reducing the risks associated with handling commodity fluoride chemicals.
Iron fouling can also be difficult to remove. Oxidized iron deposits may respond partially to acidic cleaning, but if buildup is significant, simple pH adjustment is usually not sufficient to fully dissolve the deposits. Citric acid is often assumed to be effective for iron removal because it can complex soluble iron species; however, once iron has precipitated and formed dense ferric hydroxide deposits on the membrane surface, citric acid alone typically has limited ability to dissolve and remove the fouling layer. Iron sulfide species are even more resistant and will not dissolve under standard acid or caustic conditions. In these cases, specialty formulations are often needed to solubilize and carry the iron away from the membrane.
Biological and organic fouling adds another layer of complexity. Biofilm is not just loose organic material. It is a structured matrix that, if not addressed correctly, can build up quickly and significantly impact membrane performance. Low pH can actually cause biofilm to compact and become denser, similar to how milk curdles when acid is added. High pH alone may loosen organic material, but without proper surfactants or dispersants, the deeper structure can remain.
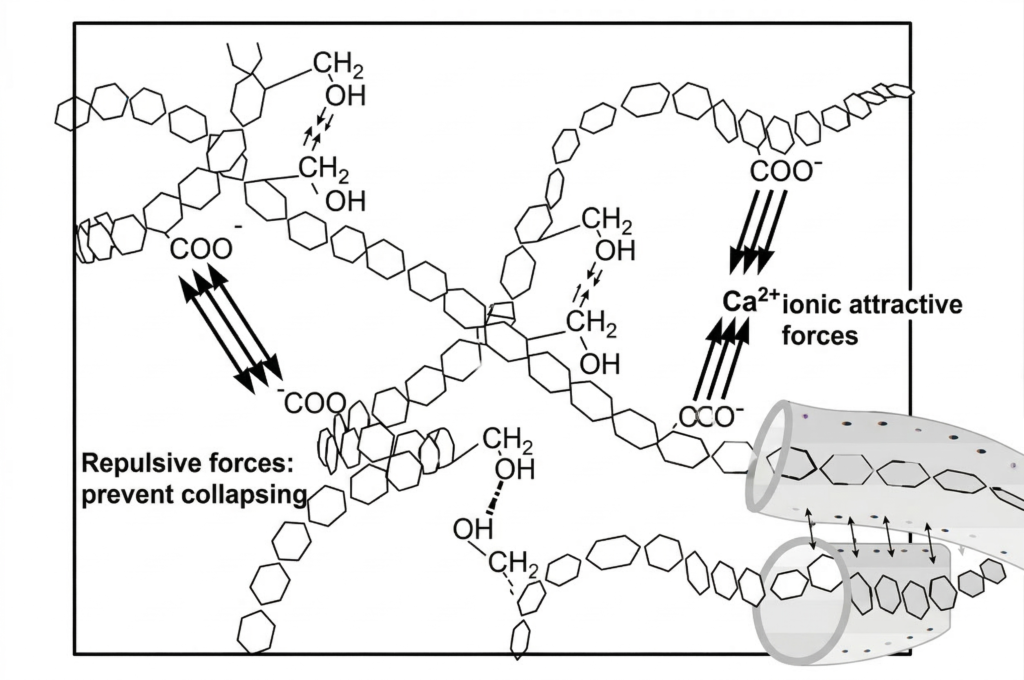
Graphic 5: Biofilm Symposium: Professor Hans-Curt Flemming, Biofilm Centre, University of Duisburg-Essen (Mayer et al., 2001)
In most systems, fouling is not just one material. Biofilm may form and act as a matrix trapping inorganic scale and suspended solids. If it’s not fully removed during a cleaning, it can protect embedded scale and make mechanical flushing even less effective. Because inorganic crystals are not negatively affected by high pH, but biofilm can become denser under low pH conditions, the cleaning sequence matters. For this reason, high pH cleaning is generally performed first, followed by acid cleaning if needed. The main exception is when it is confirmed that only inorganic scale is present.
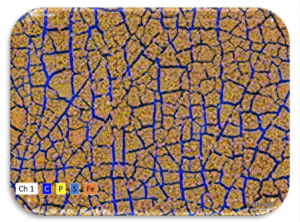
Graphic 6: Iron oxide/hydroxide embedded into a dense layer of biofilm
The key takeaway is simple: different foulants behave differently. If the chemistry does not match how the foulant behaves, the cleaning will be incomplete. Without confirming what is actually on the membrane, selecting a CIP protocol becomes guesswork rather than strategy.
Determining the Right Cleaning Protocol
Once it is understood that different foulants respond to different chemistries, the next step is to determine which protocol will effectively address the specific deposits present in the system. That process begins with confirmation.
A membrane autopsy provides the foundation. By opening a fouled element and examining the deposits, we can determine what materials are present, how they are distributed, and whether fouling is layered. The analysis may identify inorganic scale, such as calcium-based salts, iron deposits, silica, organic material, biofilm, or a combination of these. It also reveals whether fouling is primarily on the membrane surface or within the feed spacer.

Graphic 7: Membrane Autopsy
This information is critical because the cleaning strategy depends directly on what is found. For example, if both iron and biofilm are present, the protocol must address each appropriately. A high pH cleaning step may be required to break down and disperse the biological matrix, followed by a low pH step to solubilize iron deposits. The specific chemistry used in each step must be selected based on how the identified foulants behave under membrane-safe conditions.
Autopsy findings provide a starting point for developing the cleaning approach, but they do not guarantee that a proposed protocol will restore performance at full scale. That is where cleaning studies become valuable.
After an element is autopsied, membrane coupons can be cut from the fouled leaf and mounted onto laboratory test cells. Because an 8-inch element contains roughly 400 square feet of membrane surface area, multiple coupons can be collected from the same element. This allows different cleaning protocols to be tested under controlled conditions. Variables such as pH, temperature, contact time, cleaning sequence, and recirculation approach can be adjusted and compared.

Graphic 8: Flat-sheet testing skid
If the first proposed protocol does not produce satisfactory permeability recovery, additional coupons can be used to evaluate alternative conditions. This flexibility allows for cleaning chemistry and operating parameters optimization before applying them full scale. Instead of relying on assumptions, the protocol is confirmed through controlled testing.

Graphic 9: Flat-sheet cleaning study on a membrane fouled with severe organic deposits. The same cleaning procedure (pH 12.5, 35°C) was applied using a specialty organic foulant cleaner (Trial 1) and standard caustic (Trial 2). The specialty cleaner completely removed the foulant within 2 hours, while caustic showed little to no removal even after 6 hours.
One limitation of flat-sheet or coupon testing is that the feed spacer is removed during mounting. As a result, while surface fouling removal can be evaluated accurately, hydraulic performance related to spacer blockage cannot be assessed. If the system is experiencing elevated differential pressure and feed spacer fouling is suspected, a full-element cleaning study may be necessary.
In a full-element study, an intact fouled element is cleaned under realistic hydraulic conditions. Because only one full cleaning trial can be performed on a given element, this step is typically reserved for after the foulant has been identified and the chemistry has been refined through coupon testing. At this stage, the full-element study serves as confirmation that the proposed protocol restores both permeability and differential pressure under real operating conditions.
Graphic 10: Full-element cleaning study evaluating whether Cleaner B should reduce the extremely high differential pressure across the membrane. The study confirmed that the cleaner successfully restored hydraulic performance.
By progressing from autopsy to coupon testing and, when necessary, to full-element validation, a cleaning protocol can be developed specifically for the foulant present in that system. Rather than applying a standard recipe, the final recommendation is based on confirmed foulant identification and tested removal performance.
Implementing the Protocol and Confirming Results
Once the foulant has been identified and the cleaning protocol has been validated, the full-scale CIP can be performed. However, the process does not end when the system is placed back online.
The most important step after cleaning is performance verification. Normalized data must be reviewed to confirm that permeability, differential pressure, and salt rejection have returned to baseline or near-baseline values. Immediate improvement alone is not enough. The question is whether performance has been fully restored.
If recovery is incomplete, it does not necessarily mean the cleaning protocol was incorrect. In some cases, fouling may have already caused irreversible changes to the membrane that cannot be reversed by cleaning. The protocol may have successfully removed the foulant, yet the system may not fully return to its original baseline because permanent hydraulic restriction or membrane damage has already occurred. The best way to minimize this risk is to initiate cleanings when performance first deviates beyond recommended thresholds, rather than allowing fouling to persist for extended periods. Reviewing normalized data alongside autopsy and cleaning study findings helps determine whether the foulant was effectively removed or if irreversible damage has already taken place.
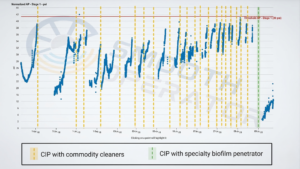
Graphic 11: Severe biofilm limited performance and standard cleanings were ineffective. After an autopsy and cleaning study determined effective protocol, a full-scale trial with a specialty biofilm penetrator restored 1st stage normalized differential pressure to baseline – the first full recovery achieved at this point.
The goal is not simply to clean when performance declines. The goal is to clean with intention, using evidence to guide chemistry selection and strategy. That way, cleanings become more predictable and more effective, intervals between cleanings extend, chemical exposure is minimized, downtime is reduced, and membrane life is preserved.
By identifying the foulant, validating the protocol, and confirming full-scale recovery, CIP moves from reactive maintenance to controlled performance management; saving time, reducing cost, and improving long-term system reliability.



